The Compound Cholesterol
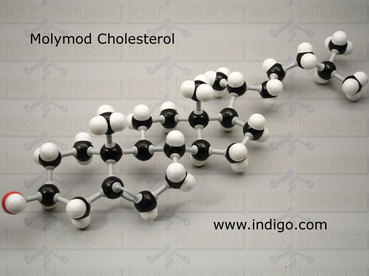
Name: Cholesterol
Formula: C27 H46 O
Molecular Mass: 386.65354 [g/mol]
Melting Point: 148–150 °C
Boiling point: 360 °C (decomposes in this state)
Density in its natural state: 1.052 g/cm^3
Where it is found: Cholesterol is found in foods and made in the liver
Natural Abundance: The liver makes about 700-900 mg a day then sends it to the call membranes. The amount of cholesterol in food varies from 0mg in corn oil to 1010 mg of cholesterol in carrot cake.
How it is obtained: Because cholesterol is made in the liver and found in the blood stream, surgery would need to occur in order to obtain cholesterol from the human body.
Type of bonding: Polar Covelant Bond
Total Mass: C= 12.0g H= 1.0g O=15.9994g
(27 x 12) + (46 x 1) + (1 x 16)= 386g
Percent composition by mass:
%C = 324/ 386 = 0.8393782383419689= 84%
%H= 46 / 386 = 0.11917098445595854= 12%
%O= 16/386= 0.04145077720207254= 4%
Formula: C27 H46 O
Molecular Mass: 386.65354 [g/mol]
Melting Point: 148–150 °C
Boiling point: 360 °C (decomposes in this state)
Density in its natural state: 1.052 g/cm^3
Where it is found: Cholesterol is found in foods and made in the liver
Natural Abundance: The liver makes about 700-900 mg a day then sends it to the call membranes. The amount of cholesterol in food varies from 0mg in corn oil to 1010 mg of cholesterol in carrot cake.
How it is obtained: Because cholesterol is made in the liver and found in the blood stream, surgery would need to occur in order to obtain cholesterol from the human body.
Type of bonding: Polar Covelant Bond
Total Mass: C= 12.0g H= 1.0g O=15.9994g
(27 x 12) + (46 x 1) + (1 x 16)= 386g
Percent composition by mass:
%C = 324/ 386 = 0.8393782383419689= 84%
%H= 46 / 386 = 0.11917098445595854= 12%
%O= 16/386= 0.04145077720207254= 4%
